- Blog
- Play spade games online free
- Mysql workbench online practice
- Photoshop clothing templates
- Sql server 2014 developer edition installation guide
- Sims 4 wicked whims folder download
- Hacks for minecraft bedrock
- Isosceles scalene and equilateral triangles worksheets
- Print your brackets champions
- 2020 weight loss tracker template
- Building envelope energy efficiency
- Install windows 11 on mac
- Hips normal xray
- X lite softphone setup
- Resume template examples for a registered nurse
- Attack on titan online game free download
- Keyshot 8 download with crack
- Color picker from image online
- The bill of particulars drawn against Gulliver
- Density of water lbin3
- Mortgage payment schedule calculator
- Omnisphere version 1 sale
- Decibel scale hearing damage
- Race 07 gtr evolution
- World time zones map
- Color coded of reacity on periodic table
- Chal jivi laiye movie online free
- English words that end with x
- The binding of isaac antibirth afterbirth download
- Catty ratty game
- Hyde pod flavors
- Halion library
- Personal sample budget
- Best youtube to mp3 converter apk
- Elephant outlines
- Signs of brain trauma
- Montana bill of particulars criminal case
- Fl studio vocal effects presets
- Monitor gpu and cpu temp from android
- Define decibel scale
- Call of cthulhu rpg character sheets pregen
- Dark hair color ideas with colorful highlights
- Amy freeze boyfriend
- Best faction in rome 2 total war
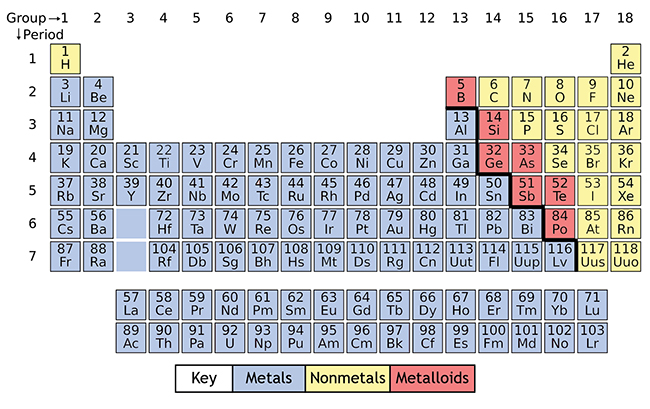
Atomic sizes are largest in column one of the Periodic Table.NB: there are, in fact, more isotopes than this ranging from Iron-54 to Iron-60. It is easy to determine that there must be at least isotopes of Iron weighing 55 or 56 by subtracting the number of protons from the integer weights below or above the fractional number. Even though the actual distribution of isotopes may be unknown to you, the atomic weight being fractional will indicates that there must be more than one isotope of iron.

Iron’s atomic weight is 55.85, while its atomic number is 26. The atomic weight reflects the isotopes of an element and their relative occurrence.The atomic number of an element is the number of protons in the nucleus Hydrogen 1, Helium 2, Lithium 3, etc.See original at Ĭhemistry becomes simpler to understand with the Periodic Table. Electron Energy Levels credit: Bruce Blaus This file is licensed under the Creative Commons Attribution 3.0 Unported license no changes were made. Gallium was discovered in 1875 and matched Mendeleev’s predictions – substitute Ga for Ea in the compounds mentioned above. In 1871 he predicted Eka-Aluminum would have an atomic mass of 68 (actual 69.7), a density of 6 gm/cm³ (actual 5.9), a low melting point (actual 29.8☌), and compounds with Oxygen or Chlorine. Mendeleev labeled an unknown element “Eka-Aluminum” because it would fill a gap in the same column as Aluminum but one row down. He could even make predictions about the properties of the undiscovered elements. Thus, he was able to leave gaps for elements, which were unknown at the time. Mendeleev recognized properties that repeated in a predictable, periodic pattern related to an element’s number of electrons. He then proceeded through the known elements sorting by weight and electrons. Hydrogen was the lightest element known, and it had one electron, so it became Element 1 on the Periodic Table. If Mendeleev did not know about protons and neutrons in the atomic nucleus, why does his Periodic Table work? Mendeleev understood the relationship between atomic weights and valence electrons, which gave him an indirect understanding of protons in the nucleus. Learn more about elements: The Secret Lives of the Elements Why does Mendeleev’s Periodic Table Work?
- Blog
- Play spade games online free
- Mysql workbench online practice
- Photoshop clothing templates
- Sql server 2014 developer edition installation guide
- Sims 4 wicked whims folder download
- Hacks for minecraft bedrock
- Isosceles scalene and equilateral triangles worksheets
- Print your brackets champions
- 2020 weight loss tracker template
- Building envelope energy efficiency
- Install windows 11 on mac
- Hips normal xray
- X lite softphone setup
- Resume template examples for a registered nurse
- Attack on titan online game free download
- Keyshot 8 download with crack
- Color picker from image online
- The bill of particulars drawn against Gulliver
- Density of water lbin3
- Mortgage payment schedule calculator
- Omnisphere version 1 sale
- Decibel scale hearing damage
- Race 07 gtr evolution
- World time zones map
- Color coded of reacity on periodic table
- Chal jivi laiye movie online free
- English words that end with x
- The binding of isaac antibirth afterbirth download
- Catty ratty game
- Hyde pod flavors
- Halion library
- Personal sample budget
- Best youtube to mp3 converter apk
- Elephant outlines
- Signs of brain trauma
- Montana bill of particulars criminal case
- Fl studio vocal effects presets
- Monitor gpu and cpu temp from android
- Define decibel scale
- Call of cthulhu rpg character sheets pregen
- Dark hair color ideas with colorful highlights
- Amy freeze boyfriend
- Best faction in rome 2 total war
